 1987), methionine sulfoxide ( Levine et al. Proteins are one of the major targets for this attack and oxidation of amino acids can generate non-native cysteine disulfideīonds ( Davies et al. However, the defense systems are not leak proof and ROS can escape and attack subcellular components, including nucleicĪcids, fatty acids, and proteins ( Halliwell and Gutteride 1984 Davies et al. Importantly, they are also produced during normal metabolic electron transport by mitochondria, chloroplasts, and respiringīacteria ( Fridovich 1978) and cells have developed antioxidant defense systems aimed at protecting against these ROS (e.g., Halliwell 1974 Demple and Halbrook 1983 Christman et al. External environmental factors including ionizing radiation and a variety of chemicals can generate ROS ( Cooper and Zika 1983 Petasne and Zika 1987). The life span of several organisms, and cancer, chronic inflammation, and cardiovascular, neurodegenerative, and pulmonaryĭiseases have been associated with age-related oxidative stress (e.g., Stadtman 1992). Reactive oxygen species (ROS) such as hydrogen peroxide, superoxide anion, and hydroxyl radicals have been implicated in determining The possibility that oxidative sensitivity of TCA cycleĮnzymes serves as a feedback mechanism down-regulating toxic respiration is discussed. In addition, cells lacking cytoplasmic superoxideĭismutase activity exhibit superinduction of heat shock proteins. That this induction is mitigated by overproducing the superoxide dismutase SodA. Stasis serves as the signal for stationary-phase, developmental, induction of the heat shock regulon is presented by demonstrating Evidence that oxidation of target proteins during Glutamine synthetase, glutamate synthase, pyruvate kinase, DnaK, and H-NS. Some proteins were found to be specifically susceptible to stasis-induced oxidation notably several TCA cycle enzymes, The stasis-induced increase in protein oxidation is enhanced in cells lacking the global regulators OxyR and In this work, we demonstrate that the levels of oxidized proteins is correlated to the age of a stationary-phase Escherichia coli culture both disulfide bridge formation of a cytoplasmic leader-less alkaline phosphatase and protein carbonyl levels increaseĭuring stasis. To all life forms, including growth-arrested bacteria, is why growing older by necessity causes organisms to grow more fragile. Finally, these results allowed to reinforce the hypothesis of oxidative damage in erythrocyte membrane proteins as molecular mechanism of human adaptation to malaria infection.Aging, or senescence, is the progressive deterioration of every bodily function over time. Therefore, both polymorphisms promote carbonylation on the same membrane proteins. 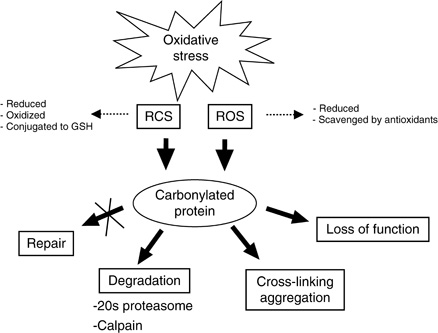
Erythrocytes with G6PD deficient and SCT showed higher carbonyl index values than control and similar profiles of carbonylated proteins moreover, cytoskeletal and stress response proteins were identified as the main targets of oxidative damage. Besides, protein carbonylation profiles were obtained by Western blot and complemented with mass spectrometry using MALDI-TOF-TOF analysis. 
Carbonyl index by dot blot assay was used to calculate the variation of oxidative damage during the asexual phases. falciparum 3D7 were used to quantify oxidative damage in membrane proteins of erythrocytes with G6PD deficient and SCT. Here, synchronous cultures at high parasitemia levels of P. Nevertheless, mechanisms are not completely understood at molecular level for each polymorphism yet, and even less if are commons for several of them. There exist in both a prooxidant environment that favors the oxidative damage on membrane proteins, which probably is part of molecular protector mechanisms. Both are considered the result of the selective pressure exerted by Plasmodium parasites over human genome, due to a certain degree of resistance to the clinical symptoms of severe malaria. Deficiency of glucose-6-phosphate dehydrogenase (G6PD) and sickle cell trait (SCT) are described as the polymorphic disorders prevalent in erythrocytes. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
